What Is NAD+? Cellular Energy, Aging Biology, and Metabolic Health
Redox Metabolism, Cellular Signaling, and Bioenergetic Capacity
NAD+ is often described as an “energy molecule” or a “longevity cofactor.”
Both descriptions capture part of its role but miss the broader biological context.
Nicotinamide adenine dinucleotide (NAD+) is a central metabolic cofactor involved in redox reactions, mitochondrial respiration, DNA repair, and cellular signaling pathways. Rather than acting as a hormone or peptide signal, NAD+ functions as a biochemical substrate that enables multiple enzymatic systems to operate.
Understanding NAD+ requires separating metabolic function from supplement narratives. NAD+ itself is not a signaling peptide like MOTS-c and it does not stabilize mitochondrial membranes like SS-31. Instead, it supports the enzymatic machinery that governs energy metabolism and cellular maintenance.
This issue explains what NAD+ is, how it functions in metabolism, how it interacts with mitochondrial biology, and how it compares mechanistically with compounds such as MOTS-c and SS-31.
What Is NAD+?
NAD+ (nicotinamide adenine dinucleotide) is a coenzyme present in every living cell.
It exists in two primary forms:
NAD+ (oxidized form)
NADH (reduced form)
This redox pair functions as an electron carrier in metabolic reactions.
NAD+ plays essential roles in:
Cellular respiration
Mitochondrial ATP production
DNA repair pathways
Cellular stress responses
Sirtuin signaling
Unlike peptides discussed in previous issues, NAD+ is not produced as a signaling molecule but as a metabolic cofactor required for energy conversion.
How NAD+ Works (Mechanism of Action)
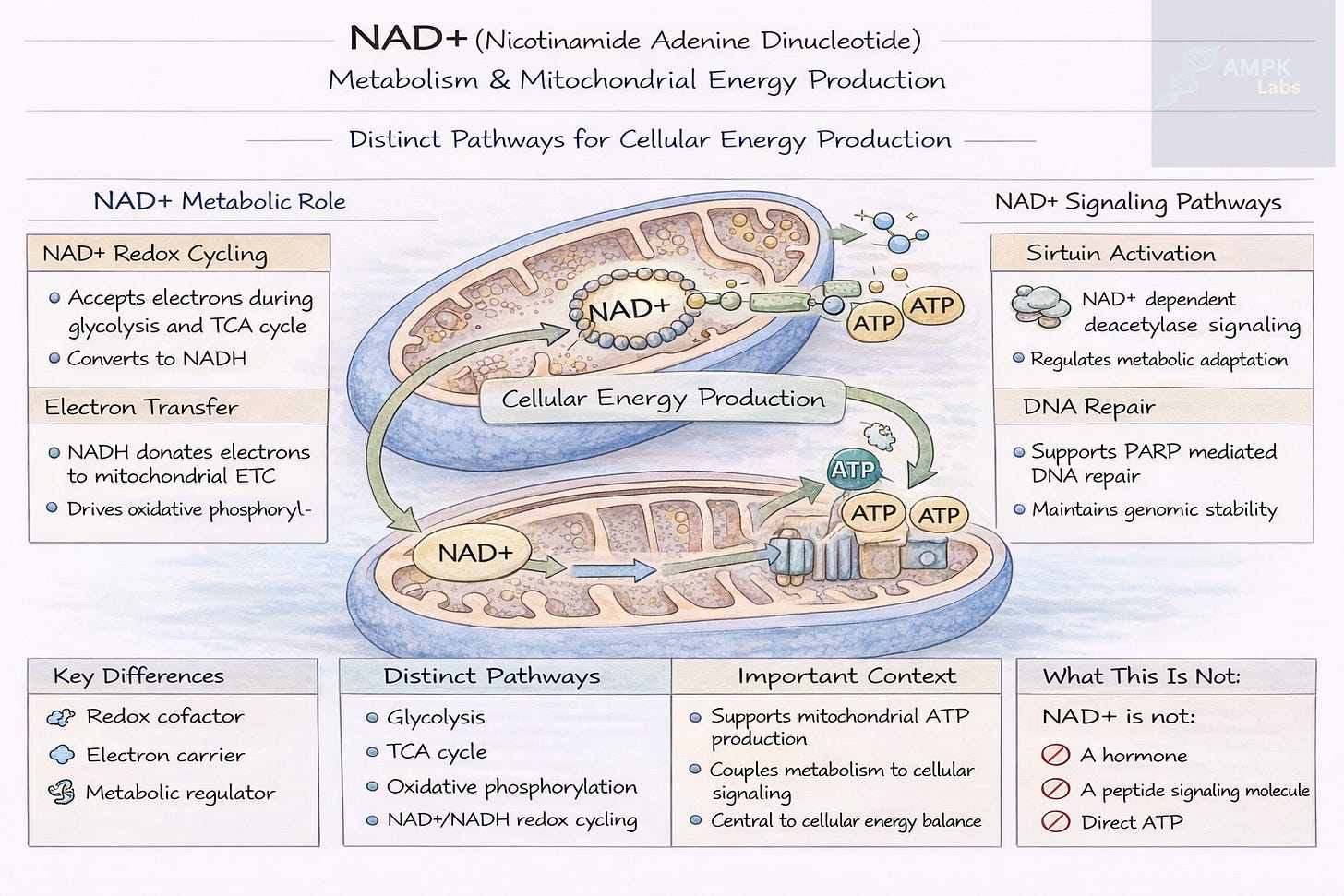
NAD+ functions primarily as an electron carrier in cellular metabolism.
During glycolysis and the tricarboxylic acid (TCA) cycle, NAD+ accepts electrons and becomes NADH. NADH then transfers those electrons to the mitochondrial electron transport chain, enabling ATP production.
Beyond redox metabolism, NAD+ also acts as a substrate for several enzyme families.
Key NAD+-dependent enzymes include:
Sirtuins (SIRT1–SIRT7)
Poly ADP ribose polymerases (PARPs)
CD38 and CD157 ectoenzymes
These systems influence:
DNA repair
Cellular stress responses
Inflammatory signaling
Metabolic regulation
Because these enzymes consume NAD+, cellular NAD+ levels influence multiple regulatory pathways simultaneously.
Educational Mechanism Diagram
NAD+ vs MOTS-c vs SS-31 (Key Differences)
NAD+
Metabolic cofactor
Electron carrier in cellular respiration
Substrate for sirtuins and DNA repair enzymes
Supports mitochondrial metabolism indirectly
MOTS-c
Mitochondrial derived peptide
Regulates metabolic stress signaling
Influences AMPK activation and nuclear gene expression
SS-31 (Elamipretide)
Mitochondria targeting tetrapeptide
Binds cardiolipin in the inner mitochondrial membrane
Stabilizes electron transport chain structure
These three molecules operate at different layers of mitochondrial biology.
NAD+ supports enzymatic metabolism.
MOTS-c regulates metabolic signaling.
SS-31 stabilizes mitochondrial architecture.
What the Research Shows
Preclinical Evidence
Cellular and animal studies demonstrate that NAD+ levels influence metabolic resilience, mitochondrial function, and stress response pathways.
Raising NAD+ availability has been shown to:
Improve mitochondrial oxidative metabolism
Enhance sirtuin activity
Influence insulin sensitivity in metabolic models
Support DNA repair mechanisms
These findings have led to interest in NAD+ biology within aging and metabolic research.
Human Evidence
Human research primarily focuses on NAD+ metabolism and its precursors, such as nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN).
Clinical studies show that NAD+ precursor supplementation can increase circulating NAD+ metabolites and influence metabolic biomarkers.
Direct intravenous NAD+ administration has also been explored in clinical settings, although mechanisms and pharmacokinetics remain an active area of investigation.
Common Public Claims and How They Compare to Evidence
NAD+ boosts cellular energy
Evidence supports that NAD+ participates directly in metabolic pathways that produce ATP.
However, NAD+ does not function as a stimulant or immediate energy enhancer.
NAD+ reverses aging
NAD+ biology is deeply connected to aging related pathways, particularly through sirtuins and DNA repair systems.
Current evidence demonstrates associations with metabolic health rather than definitive anti-aging outcomes.
NAD+ works the same as NAD precursors
NAD+, NR, and NMN participate in the same metabolic network but differ in pharmacokinetics and cellular uptake pathways.
Their biological effects cannot be assumed to be identical.
Risks and Unknowns
Important areas of ongoing research include:
Long-term effects of sustained NAD+ elevation
Interactions with sirtuin and PARP activity
Differences between precursor supplementation and direct NAD+ administration
Tissue specific NAD+ metabolism
As research evolves, these factors will clarify how NAD+ biology interacts with mitochondrial function and metabolic regulation.
Key Takeaways
NAD+ is a central metabolic cofactor required for cellular energy production
It functions as an electron carrier and enzymatic substrate
It supports mitochondrial metabolism but does not directly signal adaptation
Its role differs fundamentally from mitochondrial peptides such as MOTS-c and SS-31
It sits at the core of cellular redox biology
Sources
Rajman L, Chwalek K, Sinclair DA.
Therapeutic potential of NAD boosting molecules: the in vivo evidence.
Cell Metabolism. 2018.
https://pubmed.ncbi.nlm.nih.gov/29514064/
Xie N, Zhang L, Gao W, et al.
NAD+ metabolism: pathophysiologic mechanisms and therapeutic potential.
Signal Transduction and Targeted Therapy. 2020.
https://www.nature.com/articles/s41392-020-00311-7
Guo C, Huang Q, Wang Y, et al.
Therapeutic application of natural products: NAD+ metabolism as potential target.
Phytomedicine. 2023.
https://www.sciencedirect.com/science/article/pii/S0944711323001290
Yoshino J, Baur JA, Imai SI.
NAD+ intermediates: The biology and therapeutic potential of NMN and NR.
Cell Metabolism. 2018.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5842119/